More Information
Submitted: March 05, 2026 | Accepted: March 17, 2026 | Published: March 18, 2026
Citation: Taki H, El Mawla Z, Damen B, Raad A. When the Lungs Deceive: The Fatal Course of a Hidden Pulmonary Vein Thrombus. J Pulmonol Respir Res. 2026; 10(1): 012-015. Available from:
https://dx.doi.org/10.29328/journal.jprr.1001077
DOI: 10.29328/journal.jprr.1001077
Copyright license: © 2026 Taki H, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Pulmonary vein thrombosis; Hypoxemic respiratory failure; Right heart failure; Transesophageal echocardiography; Multi-organ failure; Case report
When the Lungs Deceive: The Fatal Course of a Hidden Pulmonary Vein Thrombus
Hanan Taki1, Zeinab El Mawla1*, Bilal Damen2 and Ali Raad1,2
11Department of Pulmonary & Critical Care, Faculty of Medical Sciences, Lebanese University, Hadat, Lebanon
2Pulmonary and Critical Care Division, Rassoul Aazam Hospital, Beirut, Lebanon
*Corresponding author: Zeinab El Mawla, Department of Pulmonary & Critical Care, Faculty of Medical Sciences, Lebanese University, Hadat, Lebanon, Email: [email protected]
Background: Pulmonary vein thrombosis (PVT) is a rare yet critically perilous vascular condition, frequently eluding timely diagnosis due to its protean clinical manifestations. While typically linked to underlying pathologies such as malignancy, thoracic surgical interventions, or atrial fibrillation ablation, PVT can, in exceptionally rare instances, manifest idiopathically. This report details an unusual case of acute respiratory failure attributable to PVT in a previously robust woman, notably devoid of conventional risk factors.
Case presentation: A 54-year-old woman, with no significant past medical or surgical history, presented to the emergency department exhibiting rapidly progressive dyspnea, profound fatigue, pleuritic chest pain, and palpitations. Upon admission, she displayed severe hypoxemia (SpO2 70% on room air), marked tachycardia, fever, and tachypnea. Comprehensive initial diagnostic evaluations, including imaging and laboratory analyses, effectively excluded more common etiologies such as pulmonary embolism, pneumonia, and COVID-19. Despite aggressive supportive measures and mechanical ventilation, her clinical course rapidly deteriorated, complicated by unstable atrial fibrillation and progressive multi-organ failure, culminating in her demise on the fifth day of hospitalization. Transesophageal echocardiography (TEE) definitively established the diagnosis of pulmonary vein thrombosis, with extension into the left atrium. Although hypercoagulability screening was initiated and systemic anticoagulation commenced, these interventions proved insufficient. The patient ultimately succumbed to intractable shock and widespread multi-organ dysfunction.
Conclusion: This compelling case profoundly highlights the imperative of including PVT in the differential diagnosis for patients presenting with severe, unexplained hypoxemia and signs of right heart failure, particularly when initial conventional imaging studies yield inconclusive results. Timely echocardiographic assessment and the immediate initiation of anticoagulation hold the potential to be life-saving, though the overall prognosis for such fulminant presentations remains exceedingly grave.
Pulmonary vein thrombosis (PVT) stands as an uncommon, yet potentially catastrophic, vascular disorder characterized by the thrombotic occlusion of one or more pulmonary veins. These vital vessels are responsible for conveying oxygenated blood from the lungs back to the left atrium of the heart. PVT is conventionally associated with specific predisposing conditions, including malignancies (especially lung cancer), thoracic surgical procedures (such as lung transplantation), radiofrequency catheter ablation for atrial fibrillation, chest trauma, or congenital vascular anomalies. However, in exceedingly rare circumstances, PVT can arise idiopathically, presenting a significant diagnostic challenge [1,2].
The clinical manifestations of PVT are often insidious and non-specific, encompassing a spectrum of symptoms from dyspnea and pleuritic chest pain to cough, hypoxemia, or hemoptysis. This inherent lack of specificity in its clinical presentation frequently contributes to misdiagnosis or considerable delays in establishing the correct diagnosis, as these features can readily overlap with more prevalent conditions like pulmonary embolism, pneumonia, or heart failure [3,4].
This case report meticulously details a fatal presentation of PVT in a previously healthy woman who rapidly developed acute respiratory failure, intractable arrhythmia, and multi-organ failure. The definitive diagnosis was confirmed post-intubation through transesophageal echocardiography (TEE), underscoring the critical role of advanced imaging in such elusive cases.
A 54-year-old woman, with no discernible past medical or surgical history, presented to the emergency department with a four-day history of progressively worsening shortness of breath, profound generalized fatigue, intermittent palpitations, and pleuritic chest pain. She explicitly denied any recent fever, productive cough, sore throat, or antecedent upper respiratory symptoms. Pertinently, there was no history of recent immobility, trauma, international travel, known malignancy, or chronic medication use.
Upon examination, the patient exhibited overt signs of acute respiratory distress. Her oxygen saturation was critically low at 70% on room air. Vital signs revealed a febrile state (38.2 °C), marked tachypnea (respiratory rate 32 breaths/min), and tachycardia (heart rate 110 bpm). Blood pressure remained within normal limits. Physical examination further revealed the prominent use of accessory respiratory muscles and jugular venous distension, though notably, there was an absence of lower limb edema or adventitious lung sounds on auscultation. Neurologically, she remained alert and oriented at presentation, with no focal deficits.
Given the severity of her hypoxia, the patient was immediately placed on high-flow oxygen via a non-rebreather mask at 15 L/min. Initial laboratory investigations revealed elevated inflammatory markers, a prolonged prothrombin time, significantly elevated D-dimer, and markedly increased troponin (1.1 ng/mL) and proBNP levels (7866 pg/mL). Renal function was initially preserved. Nasopharyngeal swabs for SARS-CoV-2 and influenza A/B yielded negative results.
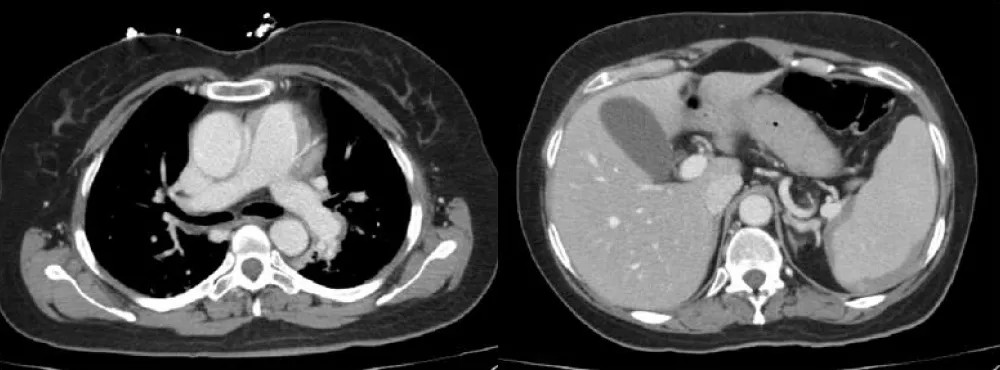
An electrocardiogram demonstrated sinus tachycardia. A chest X-ray was unremarkable, showing no infiltrates or signs of consolidation. To promptly exclude pulmonary embolism, a CT pulmonary angiogram was urgently performed. The scan revealed no evidence of pulmonary emboli, no parenchymal abnormalities, and only mild dependent atelectasis. An incidental finding of a mild subacute-chronic splenic subcapsular hematoma was noted (Figure 1).
Figure 1: CT pulmonary angiogram revealed no evidence of pulmonary emboli, with an incidental mild subacute-chronic splenic subcapsular hematoma.
Over the ensuing hours, the patient’s condition deteriorated precipitously, necessitating endotracheal intubation and mechanical ventilation. She subsequently developed rapid, unstable atrial fibrillation, which was managed with synchronized cardioversion. A transthoracic echocardiogram (TTE) was performed, followed by a transesophageal echocardiogram (TEE). The TEE provided the definitive diagnosis, revealing a large, mobile, echogenic mass measuring approximately 3.2 x 1.8 cm. This thrombus was located within the right superior pulmonary vein and extended significantly into the left atrium, partially obstructing the mitral valve inflow during diastole. The morphology was consistent with an acute thrombus rather than a chronic organized mass or myxoma.
Despite the immediate initiation of systemic anticoagulation with intravenous unfractionated heparin, the patient’s clinical status continued to worsen. She became hypotensive, requiring escalating doses of vasopressors, specifically norepinephrine, to maintain adequate mean arterial pressure. Neurologic deterioration ensued despite the cessation of sedatives, raising significant concerns for cerebral hypoperfusion. Subsequent laboratory investigations revealed a substantial decline in platelet count and compelling evidence of microangiopathic hemolytic anemia, characterized by elevated lactate dehydrogenase (LDH), low haptoglobin, and the presence of schistocytes on peripheral smear. A diagnosis of disseminated intravascular coagulation (DIC) was established, and she was treated with fresh frozen plasma.
Concurrently, the patient developed acute kidney injury, evidenced by rising creatinine levels and oliguria, consistent with evolving multi-organ failure. Repeat brain, chest, and abdominal CT scans showed no acute ischemia or hemorrhage and confirmed the stability of the previously noted splenic hematoma.
Consultations with hematology and cardiology teams were promptly initiated, and a comprehensive hypercoagulable workup was commenced, including tests for antiphospholipid antibodies, antithrombin III, protein C and S levels, and genetic thrombophilia mutations. Coronary angiography was performed due to the persistently elevated troponins and demonstrated normal coronary arteries, effectively excluding myocardial infarction as the primary cardiac insult.
Despite all intensive supportive measures, the patient’s condition continued its inexorable decline. She tragically succumbed on the fifth day of admission due to refractory cardiogenic shock and profound multi-organ failure.
The patient’s clinical course is summarized in the chronological timeline below (Table 1).
| Table 1: Chronological Timeline of Clinical Course. | ||
| Timeframe | Clinical Status & Interventions | Key Findings |
| Days 1–4 (Pre-admission) | Progressive dyspnea, fatigue, pleuritic chest pain, and palpitations. | No prior medical history or risk factors. |
| Day 1 (Admission) | Emergency Department presentation: severe respiratory distress. | SpO₂ 70% (room air), Temp 38.2°C, HR 110 bpm, RR 32 bpm. |
| High-flow oxygen (15 L/min) was initiated. | Elevated D-dimer, Troponin (1.1 ng/mL), proBNP (7866 pg/mL). | |
| CT Pulmonary Angiogram (CTPA). | Negative for PE; unremarkable lung parenchyma. | |
| Day 2 | Precipitous respiratory decline; endotracheal intubation. | Development of unstable atrial fibrillation. |
| Synchronized cardioversion; TTE followed by TEE. | TEE: Large thrombus in the right superior pulmonary vein extending into the left atrium. | |
| Initiation of IV Unfractionated Heparin. | Escalating norepinephrine requirements. | |
| Day 3 | Hemodynamic instability and neurologic deterioration. | Labs: DIC (low platelets, schistocytes, high LDH, low haptoglobin). |
| Multi-organ failure (MOF) progression. | Acute Kidney Injury (AKI) with oliguria. | |
| Day 4 | Refractory shock; continued vasopressor support. | Repeat CT brain/chest/abdomen: Stable findings, no acute hemorrhage. |
| Day 5 | Terminal decline. | Death due to refractory cardiogenic shock and multi-organ failure. |
Pulmonary vein thrombosis, though an uncommon clinical entity, carries potentially devastating consequences if its presence remains unrecognized. While PVT is well-documented in specific clinical contexts, such as post-lung transplantation (where its incidence can be as high as 15%), following catheter ablation for atrial fibrillation, and in the setting of thoracic malignancy, its occurrence in patients without such predisposing conditions is exceedingly rare. Idiopathic PVT, as exemplified by this case, is particularly unusual and remains poorly understood [1,2].
Pathophysiologically, PVT leads to an obstruction of pulmonary venous drainage, resulting in a significant increase in venous pressure within the affected lung segments. This venous congestion can progress to interstitial edema, alveolar hemorrhage, and severe impairment of gas exchange. Over time, the sustained elevation in pulmonary venous pressure can induce secondary pulmonary arterial hypertension, thereby increasing right ventricular afterload and potentially precipitating right-sided heart failure or cor pulmonale [3].
Clinically, PVT often presents with vague and non-specific symptoms—dyspnea, pleuritic chest pain, fatigue, cough, and occasionally hemoptysis or hypoxemia. The inherent lack of specificity in its clinical presentation frequently contributes to misdiagnosis or significant delays in diagnosis. In our patient, the initial presentation strikingly mimicked acute pulmonary embolism or sepsis-related acute lung injury; however, subsequent imaging studies definitively ruled out pulmonary arterial thrombus and infectious infiltrates [4].
Diagnosis of PVT relies heavily on advanced imaging modalities. While CT pulmonary angiography is routinely employed to evaluate the pulmonary vasculature, it may inadvertently miss PVT if the scan timing is not precisely optimized to capture the venous phase. Transesophageal echocardiography (TEE) remains the gold standard for directly visualizing thrombi within the pulmonary veins and the left atrium. TEE offers superior spatial resolution and enables direct visualization of the thrombus, particularly when it extends into the left atrium, as was critically observed in our case [5].
Laboratory findings in PVT are generally non-specific but may include elevated D-dimer, inflammatory markers, or indicators of end-organ dysfunction. In our patient, the elevated cardiac biomarkers and proBNP were initially interpreted as myocardial injury or demand ischemia; however, the subsequent echocardiographic findings unequivocally pointed towards significant right-sided strain secondary to PVT-induced pulmonary hypertension. The abrupt onset of atrial fibrillation with concomitant hemodynamic collapse further compounded the complexity of her clinical course [4,5].
The cornerstone of PVT treatment involves systemic anticoagulation. In the absence of contraindications, unfractionated or low-molecular-weight heparin is promptly initiated to prevent thrombus propagation and systemic embolization. In refractory cases or when anticoagulation is contraindicated, interventional thrombectomy or surgical resection may be considered. Crucially, supportive management targeting right heart failure, pulmonary hypertension, and systemic hypoperfusion is paramount. Regrettably, the late diagnosis and rapid clinical decline observed in this patient severely limited the effectiveness of anticoagulation [6].
This case also starkly illustrates the profound risk of multi-organ dysfunction associated with PVT, including disseminated intravascular coagulation (DIC), acute renal failure, and altered mental status likely resulting from systemic embolization or widespread hypoperfusion. The incidental finding of a subcapsular splenic hematoma may further corroborate the occurrence of microvascular embolic events. The rapid evolution of these severe complications, progressing from initial presentation to death within a mere five days, underscores the aggressive and fulminant nature of this condition when left untreated.
Pulmonary vein thrombosis is a rare, life-threatening condition that warrants high consideration in the differential diagnosis for patients presenting with unexplained severe hypoxemia, signs of right heart failure, or cardiogenic shock, especially after more common etiologies like pulmonary embolism and pneumonia have been excluded. Diagnostic delays are unfortunately common due to the non-specific clinical presentation. Early and targeted imaging, particularly with transesophageal echocardiography, is critical for prompt and accurate diagnosis. While immediate initiation of anticoagulation and comprehensive supportive management are essential, they may not suffice to prevent progression in fulminant cases. Clinicians must therefore maintain an exceptionally high index of suspicion for PVT in patients exhibiting rapid clinical deterioration of unclear etiology, even in the absence of classical risk factors.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review upon request by the Editor-in-Chief of this journal.
- Sundjaja JH, Bordoni B. Anatomy, thorax, lung veins. StatPearls [Internet]. 2024 Jan.
- Chaaya G, Vishnubhotla P. Pulmonary vein thrombosis: a recent systematic review. Cureus. 2017 Jan 23;9(1):e993. Available from: https://doi.org/10.7759/cureus.993
- Iqbal S, Rao SJ, Bedard N, Haas CJ. A case of chest jolts: pulmonary vein thrombus extending into the left atrium. J Community Hosp Intern Med Perspect. 2022;12(1):92-95. Available from: https://doi.org/10.55729/2000-9666.1019
- Cavaco RA, Kaul S, Chapman T, Casaretti R, Philips B, Rhodes A, et al. Idiopathic pulmonary fibrosis associated with pulmonary vein thrombosis: a case report. Cases J. 2009;2:9156. Available from: https://doi.org/10.1186/1757-1626-2-9156
- Weerakkody Y, Bell D, Baba Y. Pulmonary vein thrombosis. Radiopaedia.org. Accessed 2024 Aug 17. Available from: https://doi.org/10.53347/rID-30088
- Wu JP, Wu Q, Yang Y, Du ZZ, Sun HF. Idiopathic pulmonary vein thrombosis extending to left atrium: a case report with a literature review. Chinese Medical Journal. 2012;125(6):1197-1200. Available from: https://pubmed.ncbi.nlm.nih.gov/22613556/