More Information
Submitted: November 14, 2022 | Approved: November 23, 2022 | Published: November 24, 2022
How to cite this article: Miller D, Makhoul K, Foster A, Ul Hosna A. Chronic thromboembolic pulmonary hypertension resulting in decompensated right heart failure. J Pulmonol Respir Res. 2022; 6: 012-015.
DOI: 10.29328/journal.jprr.1001037
Copyright License: © 2022 Miller D, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Chronic thromboembolic pulmonary hypertension resulting in decompensated right heart failure
Daniel Miller, Karim Makhoul, Allison Foster and Asma Ul Hosna*
Icahn School of Medicine, Mount Sinai Queens, 159-17 Grand Central Parkway, 1st Floor, Jamaica, NY 11432, USA
*Address for Correspondence: Asma Ul Hosna, MD, Icahn School of Medicine at Mount Sinai Queens, 159-17 Grand central parkway, 1st Floor, Jamaica, NY 11432, USA, Email: [email protected]
Chronic thromboembolic pulmonary hypertension is a notoriously underdiagnosed cause of severe pulmonary hypertension. It is a form of precapillary pulmonary hypertension (PH) that results from intraluminal thrombus organization and fibrous formation which ultimately results in the complete obliteration of pulmonary arteries, resulting in increased pulmonary vascular resistance which leads to the development of pulmonary hypertension and as a result right heart failure. The mechanism involves the narrowing of the pulmonary artery which increases blood pressure within the lungs and impairs blood flow which increases the workload of the right side of the heart ultimately causing right heart failure. Pulmonary hypertension can also cause arrhythmias, blood clots, and bleeding in the lungs. Even though CTEPH is a deadly condition, among all forms of pulmonary hypertension, CTEPH is the only curable form. Echocardiography is the initial assessment tool for suspected PH. A right heart catheterization may be performed to confirm the presence of pulmonary hypertension. Confirmation of CTEPH requires a V/Q scan. Although ventilation/perfusion scintigraphy has a major role in the evaluation of patients with suspected CTEPH, nowadays CTA chest is being used widely as it produces much better-quality images compared to V/Q scan. Without treatment, the prognosis is very poor. Out of three treatment modalities such as; pulmonary endarterectomy (PEA) surgery, balloon pulmonary angioplasty (BPA), and medical therapy, surgery is the gold standard. The physician must be familiar with the disease entity, early diagnosis, and appropriate treatment to improve survival. Here we present a literature review on this topic.
Chronic thromboembolic pulmonary hypertension results from occlusion of pulmonary arteries due to the incomplete or partial resolution of pulmonary embolism which ultimately organizes as fibrotic material resulting in major pulmonary arteries obstruction [1,2]. As a result, pulmonary vascular resistance and pulmonary artery pressure (PAP) increase manifesting as pulmonary hypertension (PH), which in long term results in progressive right heart failure [2]. Pulmonary hypertension is defined as a pulmonary artery mean pressure greater than 20 mmHg or a pulmonary artery systolic pressure greater than 30 mmHg. Most cases of acute PE with treatment resolve with minimal abnormalities and normal hemodynamics. However, a small percentage of the patient population develops “post-PE syndrome”. Which presents exercise intolerance, and difficulty with daily activities resulting in quality-of-life impairments. These persistently symptomatic patients who have residual pulmonary vascular obstruction fall into two main groups: chronic thromboembolic disease (CTED) and chronic thromboembolic pulmonary hypertension (CTEPH) [3]. CTED patients have normal resting cardiopulmonary hemodynamics but there is proof of chronic pulmonary thromboembolism on imaging. On the other hand, CTEPH results from persistent obstruction of the pulmonary vasculature due to incomplete resolution of thrombus which results in pulmonary vascular re- modeling ultimately leading to pulmonary hypertension (PH) and right heart failure. CTED does not have pulmonary hypertension because of a lack of peripheral microvascular disease even though the patient has a thrombotic burden.
Even though untreated CTEPH is a deadly condition. Among all forms of pulmonary hypertension, CTEPH is the only curable form. CTEPH and/or CTED should be considered if dyspnea persists for more than 3 months after a confirmed PE diagnosis [4]. When CTEPH/CTED is suspected, a stepwise evaluation and management are needed to prevent morbidity and mortality.
CTEPH is the result of single or recurrent pulmonary emboli arising from sites of deep vein thrombosis most common sites are veins of the lower extremities. Other less common risk factors are coagulation disorder, ventriculoatrial shunt, indwelling central catheters, pacemaker leads, anti-phospholipid antibody syndrome, and cancer [5]. The natural course of most pulmonary emboli is resolution, either complete or partial after treatment. In some cases, minimal, organized residual abnormalities with the restoration of normal pulmonary function are noticed [6].
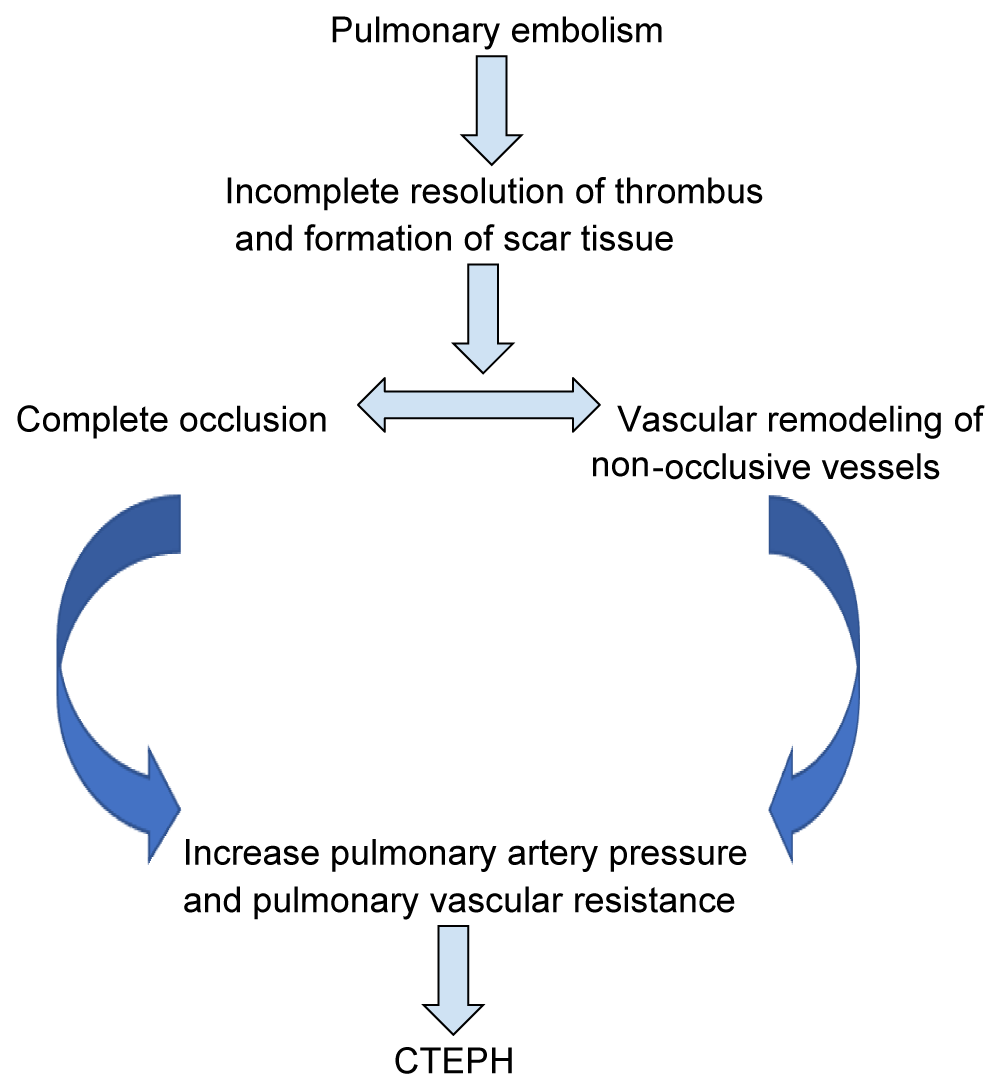
Numerous algorithms have been used to evaluate pulmonary hypertension, and all of these algorithms recommended to use ventilation/perfusion scan as a preferred screening tool for thromboembolic disease [7-9]. Confirmation of the diagnosis is done by digital subtraction pulmonary angiography. Surgical pulmonary thromboendarterectomy (PTE) is the only potentially curative option. In non-operable disease or residual disease after PTE, interventional balloon pulmonary angioplasty and/or pulmonary-vasodilator therapies can be offered, in collaboration with interventional and vascular pulmonary colleagues. As it is a disease that can cause high morbidity and mortality, CTEPH requires a high index of suspicion to diagnose and treat patients following PE [10]. In this article, we review the current state of understanding of CTEPH, diagnosis, treatment, and prognosis Figure 1.
Figure 1: Current Hypothesis about the development of CTEPH due to pulmonary embolism.
If CTEPH develops, incomplete resolution of the emboli results in either complete obstruction or significant narrowing of the central pulmonary vessels by forming fibrous scars. Which causes an increase in right ventricular (RV) afterload, right ventricular dilation, and right heart failure. The pathology behind this incomplete resolution is not clear but the combined results of age, the extent of embolus extension, coexisting pulmonary disease, and compliance with anticoagulation play an important role in the process [6]. The estimated incidence of CTEPH after acute pulmonary thromboembolism is 2% - 4% [7].
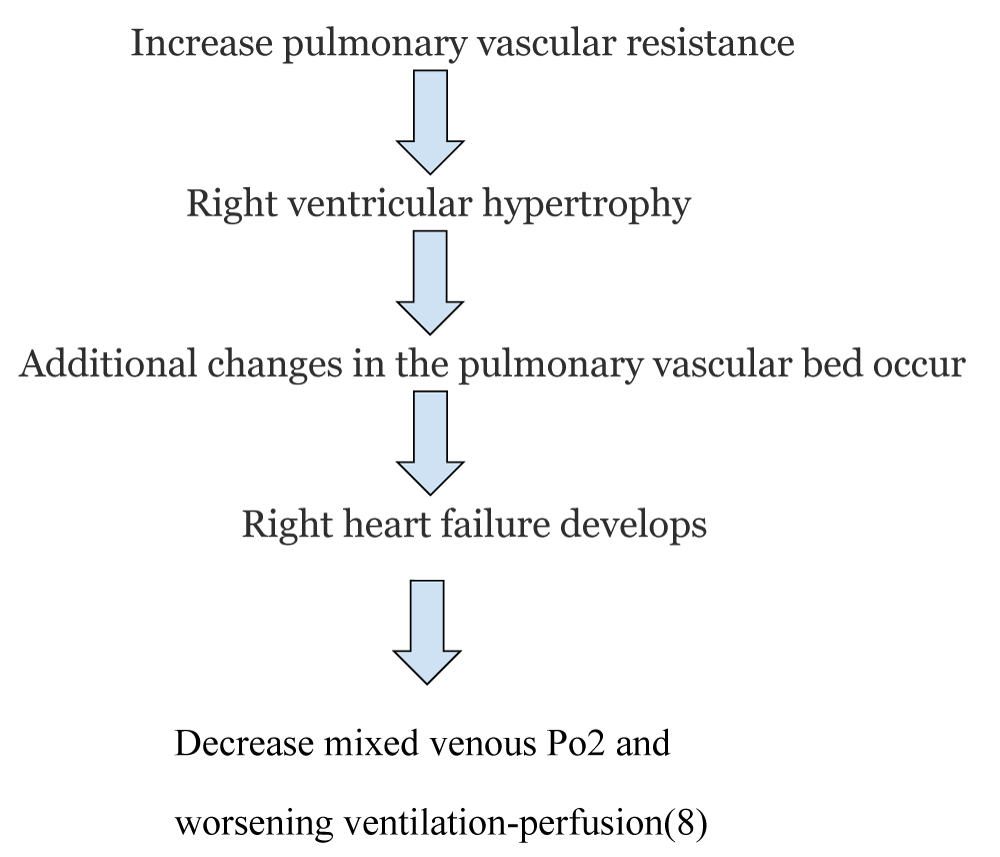
After treatment of PE with anticoagulation some people improve, initially, they develop severe dyspnea which gradually improves. Whether or not the patient receives anticoagulation for this episode, symptoms improve, and the patient returns to normal activities. This is known as the honeymoon period. Rarely need anticoagulation or follow-up lung scans [8]. The honeymoon period varies from person to person. After the honeymoon period ends, most patient starts experiencing hypoxia as a result of decompensated right ventricular dysfunction Figure 2.
Figure 2: Development of right heart failure due to CTEPH.
CTEPH should be considered if dyspnea persists for more than 3 months after a confirmed PE diagnosis [9]. When CTEPH is suspected, a stepwise evaluation and management are needed.
If the patient has signs and symptoms of pulmonary hypertension despite 3 months of anticoagulation following acute PE, Transthoracic Echocardiogram and 6 minutes walk test should be done. Echocardiographic findings are right ventricular dilatation resulting in tricuspid regurgitation (TR) which presents as tricuspid valve peak systolic gradient of more than 60 mm Hg, if evidence of pulmonary hypertension is evident, the next step is a V/Q scan. A negative V/Q can effectively rule out pulmonary embolism, V/Q scintigraphy is more sensitive than CTAP in detecting CTEPH [10]. But V/Q scan has some limitations in determining the exact location, burden, and extent of pulmonary embolism which are very important information in figuring out the appropriate management plan. So, if a mismatched segmental defect is noticed in the V/Q scan, computed tomographic angiography is done for further evaluation. CT-AP reliably identifies major vessels clot but it has limitations in diagnosing distal vessels clot. That’s why it is not the initial diagnostic tool for PE [10]. If computed tomographic angiography shows suspicion of pulmonary hypertension, the next step is to refer to the CTEPH center for right heart catheterization with pulmonary angiogram +/- CT angiogram to quantify the degree of pulmonary hypertension as well as to assess responsiveness to vasodilator. If vasodilators respond by resulting in a reduction of pulmonary artery pressure, it indicates that for the patient who undergoes pulmonary endarterectomy, the long-term survival rate will increase [11].
Once the disease is confirmed, the patient showed be assessed for operability. Right heart catheterization and CT pulmonary angiography are the gold standards for the assessment of operability [12]. If the patient is a Candidate for surgery, pulmonary endarterectomy is the treatment of choice. Even though it is a complicated procedure, it increases the quality of life of the patient. Studies have shown that after a successful thromboendarterectomy, the patient’s condition improves significantly resulting in decreasing pulmonary vascular resistance and as a result improving cardiac output [13]. Complication after PEA includes reperfusion pulmonary edema, pulmonary artery steal syndrome, residual pulmonary hypertension, and recurrent thromboembolism [14].
Pharmacological management +/-balloon angioplasty should be offered for patients who are not candidates for surgery or recurrent pulmonary embolism despite PEA.
Pharmacological management consists of diuretics, Riociguat, lifelong anticoagulation therapy, and oxygen therapy for heart failure and/or hypoxemia [15]. Endothelin receptor antagonists such as Ambrisentan and bosentan improve exercise tolerance in patients with PAH [16]. Studies showed Riociguat was effective and well-tolerated in patients of advanced age or risk factors for HFrEF. The long-term extension study demonstrated that the use of Riociguat is safe to use for up to 3 years after initiation [17]. Adverse effects of RIociguatincludes hypotension, dyspnea, dyspepsia, headaches, and upper respiratory tract infection. Macitentan also showed benefits in decreasing PVR. Epoprostenol is also very effective as it works as a pulmonary and systemic vasodilator, resulting in decreasing pulmonary vascular resistance and increasing oxygen delivery to the body [18,19]. In cases of inoperable CTEPH, Balloon pulmonary angioplasty is a promising alternative to PEA [20]. Balloon pulmonary angioplasty (BPA) has shown some benefits in both hemodynamic and functional improvement [21]. It improves right ventricular function, causes myocardial remodeling, and alleviates dyssynchrony, as well as improves left ventricular function. By improving pulmonary vascular resistance, BPA helps in the reduction of troponin-T levels, suggesting attenuation of myocardial injury. BPA also helps in improving respiratory function and oxygenation [22,23]. Even though normal pulmonary function cannot be restored fully, early treatment can prevent permanent damage to pulmonary vasculature and thus prevent the development of decompensated heart failure.
CTEPH is the only type of pulmonary hypertension that can be cured with appropriate management. But CTEPH diagnosis is delayed because of poor knowledge of the association between CTEPH with acute PE. So proper history taking along with further investigation if there is any suspicion for CTEPH is crucial. Our case has proven that delayed diagnosis or treatment can result in decompensated heart failure.
- Lang IM, Campean IA, Sadushi-Kolici R, Badr-Eslam R, Gerges C, Skoro-Sajer N. Chronic Thromboembolic Disease and Chronic Thromboembolic Pulmonary Hypertension. Clin Chest Med. 2021 Mar;42(1):81-90. doi: 10.1016/j.ccm.2020.11.014. Epub 2021 Jan 12. PMID: 33541619.
- Memon HA, Lin CH, Guha A. Chronic Thromboembolic Pulmonary Hypertension: Pearls and Pitfalls of Diagnosis. Methodist Debakey Cardiovasc J. 2016 Oct-Dec;12(4):199-204. doi: 10.14797/mdcj-12-4-199. PMID: 28289494; PMCID: PMC5344469.
- Papamatheakis DG, Poch DS, Fernandes TM, Kerr KM, Kim NH, Fedullo PF. Chronic Thromboembolic Pulmonary Hypertension: JACC Focus Seminar. J Am Coll Cardiol. 2020 Nov 3;76(18):2155-2169. doi: 10.1016/j.jacc.2020.08.074. PMID: 33121723.
- Simonneau G, Torbicki A, Dorfmüller P, Kim N. The pathophysiology of chronic thromboembolic pulmonary hypertension. 2017. 26: 160112. https://err.ersjournals.com/content/26/143/160112.figures-only
- Wilkens H, Lang I, Behr J, Berghaus T, Grohe C, Guth S, Hoeper MM, Kramm T, Krüger U, Langer F, Rosenkranz S, Schäfers HJ, Schmidt M, Seyfarth HJ, Wahlers T, Worth H, Mayer E. Chronic thromboembolic pulmonary hypertension (CTEPH): updated Recommendations of the Cologne Consensus Conference 2011. Int J Cardiol. 2011 Dec;154 Suppl 1:S54-60. doi: 10.1016/S0167-5273(11)70493-4. PMID: 22221974.
- Papamatheakis DG, Poch DS, Fernandes TM, Kerr KM, Kim NH, Fedullo PF. Chronic Thromboembolic Pulmonary Hypertension: JACC Focus Seminar. J Am Coll Cardiol. 2020 Nov 3;76(18):2155-2169. doi: 10.1016/j.jacc.2020.08.074. PMID: 33121723.
- Ozsu S, Cinarka H. Chronic thromboembolic pulmonary hypertension: Medical treatment. Pulm Circ. 2013 Apr;3(2):341-4. doi: 10.4103/2045-8932.114761. PMID: 24015333; PMCID: PMC3757827.
- Moser KM, Auger WR, Fedullo PF. Chronic major-vessel thromboembolic pulmonary hypertension. Circulation. 1990 Jun;81(6):1735-43. doi: 10.1161/01.cir.81.6.1735. PMID: 2188751.
- Barnikel M, Kneidinger N, Arnold P, Waelde A, Behr J, Milger K. Riociguat in Patients with CTEPH and Advanced Age and/or Comorbidities. J Clin Med. 2022 Feb 18;11(4):1084. doi: 10.3390/jcm11041084. PMID: 35207354; PMCID: PMC8877433.
- Tunariu N, Gibbs SJ, Win Z, Gin-Sing W, Graham A, Gishen P, Al-Nahhas A. Ventilation-perfusion scintigraphy is more sensitive than multidetector CTPA in detecting chronic thromboembolic pulmonary disease as a treatable cause of pulmonary hypertension. J Nucl Med. 2007 May;48(5):680-4. doi: 10.2967/jnumed.106.039438. PMID: 17475953.
- Skoro-Sajer N, Hack N, Sadushi-Koliçi R, Bonderman D, Jakowitsch J, Klepetko W, Hoda MA, Kneussl MP, Fedullo P, Lang IM. Pulmonary vascular reactivity and prognosis in patients with chronic thromboembolic pulmonary hypertension: a pilot study. Circulation. 2009 Jan 20;119(2):298-305. doi: 10.1161/CIRCULATIONAHA.108.794610. Epub 2008 Dec 31. PMID: 19118254.
- Doğan H, de Roos A, Geleijins J, Huisman MV, Kroft LJ. The role of computed tomography in the diagnosis of acute and chronic pulmonary embolism. Diagn Interv Radiol. 2015 Jul-Aug;21(4):307-16. doi: 10.5152/dir.2015.14403. PMID: 26133321; PMCID: PMC4498425.
- Madonna R, Cocco N, De Caterina R. Pathways and Drugs in Pulmonary Arterial Hypertension - Focus on the Role of Endothelin Receptor Antagonists. Cardiovasc Drugs Ther. 2015;29(5):469-79. doi: 10.1007/s10557-015-6605-6. PMID: 26145170.
- Madani MM. Surgical Treatment of Chronic Thromboembolic Pulmonary Hypertension: Pulmonary Thromboendarterectomy. Methodist Debakey Cardiovasc J. 2016 Oct-Dec;12(4):213-218. doi: 10.14797/mdcj-12-4-213. PMID: 28289496; PMCID: PMC5344471.
- Cheng JW, Tonelli AR, Pettersson G, Krasuski RA. Pharmacologic management of perioperative pulmonary hypertension. J Cardiovasc Pharmacol. 2014 Apr;63(4):375-84. doi: 10.1097/FJC.0000000000000050. PMID: 24710471; PMCID: PMC3982201.
- Galiè N, Olschewski H, Oudiz RJ, Torres F, Frost A, Ghofrani HA, Badesch DB, McGoon MD, McLaughlin VV, Roecker EB, Gerber MJ, Dufton C, Wiens BL, Rubin LJ; Ambrisentan in Pulmonary Arterial Hypertension, Randomized, Double-Blind, Placebo-Controlled, Multicenter, Efficacy Studies (ARIES) Group. Ambrisentan for the treatment of pulmonary arterial hypertension: results of the ambrisentan in pulmonary arterial hypertension, randomized, double-blind, placebo-controlled, multicenter, efficacy (ARIES) study 1 and 2. Circulation. 2008 Jun 10;117(23):3010-9. doi: 10.1161/CIRCULATIONAHA.107.742510. Epub 2008 May 27. PMID: 18506008.
- van Thor MCJ, Ten Klooster L, Snijder RJ, Post MC, Mager JJ. Long-term clinical value and outcome of riociguat in chronic thromboembolic pulmonary hypertension. Int J Cardiol Heart Vasc. 2019 Feb 28;22:163-168. doi: 10.1016/j.ijcha.2019.02.004. PMID: 30859124; PMCID: PMC6396196.
- Ghofrani HA, Simonneau G, D'Armini AM, Fedullo P, Howard LS, Jaïs X, Jenkins DP, Jing ZC, Madani MM, Martin N, Mayer E, Papadakis K, Richard D, Kim NH; MERIT study investigators. Macitentan for the treatment of inoperable chronic thromboembolic pulmonary hypertension (MERIT-1): results from the multicentre, phase 2, randomised, double-blind, placebo-controlled study. Lancet Respir Med. 2017 Oct;5(10):785-794. doi: 10.1016/S2213-2600(17)30305-3. Epub 2017 Sep 11. PMID: 28919201.
- Mohammadi A, Matos WF, Intriago C, Thakkar K, Jahan N, Shah H, Nishu RI, Marzban S. Use of Epoprostenol in the Treatment of Pulmonary Arterial Hypertension. Cureus. 2021 Sep 22;13(9):e18191. doi: 10.7759/cureus.18191. PMID: 34707962; PMCID: PMC8533662.
- Ikeda N. Balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension. Cardiovasc Interv Ther. 2020 Apr;35(2):130-141. doi: 10.1007/s12928-019-00637-2. Epub 2019 Dec 23. PMID: 31873853.
- Kalra R, Duval S, Thenappan T, Raveendran G, Pritzker M, Prisco SZ, Prins KW. Comparison of Balloon Pulmonary Angioplasty and Pulmonary Vasodilators for Inoperable Chronic Thromboembolic Pulmonary Hypertension: A Systematic Review and Meta-Analysis. Sci Rep. 2020 Jun 1;10(1):8870. doi: 10.1038/s41598-020-65697-4. PMID: 32483219; PMCID: PMC7264327.
- Lang I, Meyer BC, Ogo T, Matsubara H, Kurzyna M, Ghofrani HA, Mayer E, Brenot P. Balloon pulmonary angioplasty in chronic thromboembolic pulmonary hypertension. Eur Respir Rev. 2017 Mar 29;26(143):160119. doi: 10.1183/16000617.0119-2016. Erratum in: Eur Respir Rev. 2017 Jun 14;26(144): PMID: 28356406; PMCID: PMC9489135.
- Jin Q, Zhao ZH, Luo Q, Zhao Q, Yan L, Zhang Y, Li X, Yang T, Zeng QX, Xiong CM, Liu ZH. Balloon pulmonary angioplasty for chronic thromboembolic pulmonary hypertension: State of the art. World J Clin Cases. 2020 Jul 6;8(13):2679-2702. doi: 10.12998/wjcc.v8.i13.2679. PMID: 32742980; PMCID: PMC7360712.